Federal Judge Voids FDA Rule Barring Minors From “Morning After” Pill
A Federal Judge has stepped into a Culture War minefield, but that doesn't mean he's wrong.
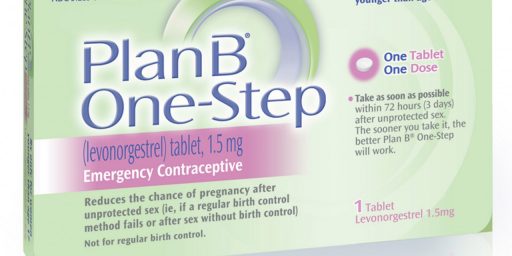
A Federal District Court Judge in New York has issued an order overturning a rule promulgated by the Food And Drug Administration mandating that girls under 17 can only receive “Plan B” and other similar “Morning After” contraceptives via prescription and ordered the FDA to allow women of any age to receive such medication over the counter:
A federal judge ruled Friday that the government must make the most common morning-after pill available over the counter for all ages, instead of requiring a prescription for girls 16 and younger. In his ruling, he also accused the federal government of “bad faith” in dealing with the requests to make the pill universally available, and said its actions had been politically motivated.
The decision, on a fraught and politically controversial subject, comes after a decade-long fight over who should have access to the pill and under what circumstances. And it counteracts an unprecedented move by the Obama administration’s Health and Human Services secretary, Kathleen Sebelius, who in 2011 overruled a recommendation by the Food and Drug Administration to make the pill available for all ages without a prescription.
In a decision in a lawsuit filed by advocates, the judge, Edward R. Korman of Federal District Court, ruled that the government’s refusal to lift restrictions on access to the pill was “arbitrary, capricious, and unreasonable.”Judge Korman ordered the F.D.A. to lift any age and sale restrictions on the pill, Plan B One-Step, and its generic versions, within 30 days.
“More than 12 years have passed since the citizen petition was filed and 8 years since this lawsuit commenced,” the judge wrote. “The F.D.A. has engaged in intolerable delays in processing the petition. Indeed, it could accurately be described as an administrative agency filibuster.”
He added, “The plaintiffs should not be forced to endure, nor should the agency’s misconduct be rewarded by, an exercise that permits the F.D.A. to engage in further delay and obstruction.”(…)
Scientists, including those at the F.D.A., have been recommending unrestricted access for years, as have major medical groups, including the American Medical Association, the American Congress of Obstetricians and Gynecologists and the American Academy of Pediatrics. They contend that the restrictions effectively keep many adolescents and younger teenagers from being able to use a safe drug in a timely way to prevent pregnancy, which carries greater safety risks than the morning-after pill.
In 2011, the F.D.A. commissioner, Dr. Margaret A. Hamburg, issued a statement saying that after rigorous study, it was safe to sell Plan B One-Step over the counter for all ages. But she was overruled by Ms. Sebelius, the Health and Human Services secretary, the first time such a public countermanding had ever occurred.
In her decision, Ms. Sebelius said that Plan B’s manufacturer had failed to study whether the drug was safe for girls as young as 11, about 10 percent of whom are physically able to bear children. But her decision was widely interpreted in a political context because emergency contraception has become an issue in the abortion debate and because allowing freer access to adolescents would prompt critics to accuse the Obama administration of supporting sexual activity for girls of that age. At the time, President Obama supported Ms. Sebelius’s decision, saying, “I will say this, as the father of two daughters: I think it is important for us to make sure that we apply some common sense to various rules when it comes to over-the-counter medicine.”
I’ve seen a lot of discussion online already about this decision, much of it quite obviously not from people who either have not read the decision or don’t really understand the law that’s involved here. Essentially, this case involves a review of rulemaking by a regulatory branch by a Federal Court pursuant to the Administrative Procedure Act, a law passed in 1946 to establish a standard for judicial review of the myriad of rules being drafted by the regulatory agencies that came into being during the New Deal and World War II. Under the law, a Federal Court must uphold a rule adopted by a Federal agency unless there is a finding, made after a hearing on the merits, that the rule is “”arbitrary and capricious, an abuse of discretion, or otherwise not in accordance with the law.”
In the case we’re dealing with here, the question is whether or not the FDA decision to limit over-the-counter availability of Plan B and related drugs to women over 17 had a rational scientific basis, or whether it was based on something other than the scientific criteria that the FDA is required by statute to consider when it’s making a decision like this. In other words, this case was not about whether or not it is a good idea as a matter of public policy to allow minors to have access to Plan B and other “morning after” drugs, but about whether a rule from the FDA that barred such access could be justified under the rulemaking authority granted to the agency. The judge’s opinion, which I’ve embedded below, goes through a rather detailed examination of both the applicable law and the scientific evidence that was presented to the Court, but it’s really the opinion’s closing paragraphs that sum up the Judge’s decision:
The decisions of the Secretary with respect to Plan B One-Step and that of the FDA with respect to the Citizen Petition, which it had no choice but to deny, were arbitrary, capricious, and unreasonable. I decline to direct a remedy comparable to that which I directed in my 2009 opinion, such as directing that emergency contraception be made available without a prescription but with the current point-of-sale restrictions to women whom studies have demonstrated are capable of understanding the label and using the product appropriately. As I have previously observed, the obstructions in the path of those adolescents in obtaining levonorgestrel-based emergency contraceptives under the current behind-the-counter regime have the practical effect of making the contraceptives unavailable without a doctor’s prescription. Consequently, the decision of the FDA denying the Citizen Petition is reversed, and the case is remanded to the FDA with the instruction to grant the Citizen Petition and make levonorgestrel-based emergency contraceptives available without a prescription and without point-of-sale or age restrictions within thirty days. On remand, the FDA may determine whether any new labeling is reasonably necessary. Moreover, if the FDA actually believes there is any significant difference between the one- and two-pill products, it may limit its over-the-counter approval to the one-pill product.
(…)
[E]ven if the defendants’ arguments would be sufficient to carry the day in the runof-the-mill case, the bad faith that has permeated consideration of the Citizen Petition, not to speak of the Plan B sponsor’s applications, should rule out such relief here. More than twelve years have passed since the Citizen Petition was filed and eight years since this lawsuit commenced. The FDA has engaged in intolerable delays in processing the petition. Indeed, it could accurately be described as an administrative agency filibuster. Moreover, one of the devices the FDA has employed to stall proceedings was to seek public comment on whether or not it needed to engage in rulemaking in order to adopt an age-restricted marketing regime. After eating up eleven months, 47,000 public comments, and hundreds of thousands, if not millions, of dollars, it decided that it did not need rulemaking after all. The plaintiffs should not be forced to endure, nor should the agency’s misconduct be rewarded by, an exercise that permits the FDA to engage in further delay and obstruction.
It was in the end a rather scathing vilification of the FDA’s actions in this matter and, reading through the opinion, one gets the clear impression that the Judge came to the conclusion that the decision by HHS Secretary Sebelius to issue the order limiting over-the-counter sales to women over 17 was motivated more by politics than by the scientific standards that the FDA and the HHS is supposed to be governed by, especially given the fact that this is an issue that has been pending for twelve years, including four years during which the Obama Administration has been in control of the relevant agencies.
I don’t consider myself enough of an expert in either the science of this matter or Federal Regulatory Law to be able to say whether Judge Edward Herman, who was appointed to the Eastern District of New York by President Reagan way back in 1985, got this matter correct. It also seems quite apparent that this decision will be appealed to the 2nd Circuit Court of Appeals, and that Judge Herman’s decision will be stayed while this appeal is pending. So, this is far from the final say on this matter. That said, to the extent that Judge Herman’s decision is based upon the factual findings he made during the course of this matter, it’s going to be very difficult to overturn the decision on appeal unless the appellate court in question finds that the court was manifestly wrong in its factual conclusions. This may make it difficult for an appellate court to overturn the decision.
Of course, most people who hear about this decision won’t be thinking about the discrete legal matters at issue in this case. They’ll be thinking about the policy question of whether minors should be able to have access to these types of medication without a prescription and, potentially, without parental knowledge and consent. As I noted above, that is a policy issue that was not within the purview of the FDA to begin with. Indeed, even if this ruling stands, it would still be within the authority of Congress to reverse the ruling via legislation that says that minors must have a prescription before obtaining the “morning after” pill. Personally, as I’ve said before, I think all contraceptives, including “morning after” pills should be available over-the-counter, but I do have some doubts about how much that policy ought to apply to minors, especially if we’re talking about girls as young as 13 or 14 years old. I’ll admit I don’t have a scientific basis for these doubts, it’s really more of a visceral reaction and a reflection of how I’d feel if we were talking about a child of mine. I’m not sure, though, that those doubts, or the fears of many on the right that their children may be more sexually active than they’d like to pretend that they are, ought to be the basis for public policy.
Here’s the opinion:




If only we could get more federal district court judges to find, well, honestly, that pretty much every other federal enforcement agency’s rulemaking was “arbitrary and capricious” and therefore invalid we’d be in a lot better shape. I’m looking at you, EPA, and and you too, HUD, NLRB, EEOC and SEC. Shizzle, speaking of HHS, if their regulatory nightmare concerning Obamacare could be tossed out on the grounds of it being arbitrary and capricious we might avoid sinking further into being a de facto economic banana republic.
Alas, when it comes to environmental regulations and housing regulations and broker-dealer regulations and wage & hour law regulations, and the whole other alphabet soup of federal regulations, getting district judges to toss them out is like pulling teeth without anesthesia. C’est la vie.
In any event, obviously it’s beyond retarded to have 12 and 13 year-old girls walking into Rite-Aid and obtaining OTC the morning after pill. Even Congress would be able to figure that out. So there could be a legislative response to this. NARAL, NOW and Emily’s List, however, for obvious reasons, vehemently will lobby against any such corrective measures. Elements of the liberal media and the academe too.
These birth control drugs have been under heavy political management for decades. The inaction under Bush was grossly theocratic and repugnant. The cowardice displayed under the Obama administration was outrageous.
Are you serious?
I mean…that’s just nonsensical.
If your opinions are based on BS…then your opinions are probably BS.
@Tsar Nicholas:
More or less retarded than having a bunch of pregnant 12 or 13 year old girls, because they couldn’t get access to Plan B?
@Tsar Nicholas: You didn’t read a single word of what Doug wrote, did you?
@legion: I kinda think Tsar is a bot that does enough text recognition of an OTB post to incorporate some very basic aspects into a machine-generated response that must include some or all of “banana republic,” “West Europe with higher crime,” “academic-media cabal,” and a host of lesser-but-still-frequent buzzwords.
If a girl as young as 13 or 14 wants a morning after pill, then that means she’s already had sex. So any concerns about keeping the precious flower innocent are pretty much out the window at that point. So then the only concern is whether you’d prefer to have a pregnant or a not pregnant 13 year old.
Ordinarily, I would think this might be a a situation where Congress should act , but I wouldn’t trust the current House Republicans to pass sensible legislation about finding your a$$, so… I say that if the decision stands, the FDA should have another go at crafting an appropriate rule. FWIW, a rule barring unlimited access to girls under 17 seems OK with me. Maybe they can do something where some caring adult can make the decision on their behalf.
I understand why this ruling gives the hives to parents, but on the other hand: the age of puberty has gotten younger and younger. How do we deal with the ramifications?
Also, is there any data on how many 13 or 14 year olds have needed Plan B? I think you’re more likely to get those just below the 17-year cutoff.
“I’ll admit I don’t have a scientific basis for these doubts, it’s really more of a visceral reaction and a reflection of how I’d feel if we were talking about a child of mine.”
Ah, the siren song of the libertarian: I believe in freedom for all, except where it vaguely bothers me for emotional reasons I can’t really articulate.
Is there a single self-styled libertarian out there who believes in freedom for others where it actually inconveniences him? I’ve sure never seen one.
@grumpy realist:
I might seem a little old fashioned, but these days to me, 13 year old girls look like 16 year old girls in my day and 16 year old girls look like women, so I have a feeling that yeah, quite a few 13 and 14 year olds might end up in Plan B situations.
I know of at least one case where a a rather mature looking 13 year old got pregnant. The father wanted her to have an abortion. Maybe if she had Plan B access, she could have taken care of the situation before her parents found out she was pregnant.
Call me old fashioned again, but I still don’t think 13 and 14 year old girls should making those kind of decisions on their own.
@grumpy realist: In addition the age of marriage has gotten later and later. In the not too distant past 14 year old girls got married and in some societies that is still the case. It’s hard to battle biology.
@Tsar Nicholas: Usually I find something in your comment that I disagree with, but not here.
@Doug @the top: A thought exercise: Do you have the same gut vicerscal reaction to the idea of a 13 year old son beng able to get condoms over the counter?
@Tsar Nicholas: Wait, so you think tha the SEC’s problem is that it does too much? Seriously?
@Mikey: I’m starting to think that TN is a bot of one of the OTB editors, designed to increase page views through idiocy.
@stonetools: What if the appropriate (in most circumstnaces) adults are the ones causing or abetting the problem? You run into the same circumstances (abusive stepfathers, willingly blind mothers, etc.) that are problematic when it comes to parental notification laws for abortion. And, since Plan B has a strict time limit on efficacy, since it is birth control and not an abortion drug, there probably really isn’t time for some sort of third party (judge, social worker) to weigh in.
@Gromitt Gunn:
I would.
I understand there are problems all around, but dagnabit… Thirteen? at thirteen I sure wasn’t equipped to make any kind of decisions like that!
OTOH
A woman told me that her mother handled the whole issue by putting her on the pill after she turned twelve. That does take all the tension out of the “Is she having sex or not? ” question. Of course, still doesn’t help with VD/AIDS but it does take pregnancy off the table.
The issue with drugs and alcohol isn’t what happens so much when they are used as intended and/or moderately with appropriate restraint, as it is what happens when they are abused, which is why it is inappropriate for the government (and judges) to reach around the family structure to provide drugs and alcohol to non-emancipated minor children. Providing open access to mature adults is another matter.
@stonetools:
The decision HAS ALREADY BEEN MADE — the girl has had sex.
Now, at this point, the only thing to do is decide how to deal with the consequences of that decision: (a) morning-after pill right away, (b) abortion some time later, (c) full-on pregnancy and then birth, or, as some here would wish, (d) crossing ones fingers and hoping the whole icky thing just goes away.
@Let’s Be Free:
Do you know many drugs and medications and medical procedures this girl is going to be subject to if she gets pregnant? It’ll make one dose of a morning-after pill look like a sugar cube.
@Rafer Janders:
I’m betting you don’t have a 13 year old daughter/daughter in-law/cousin, right? Bit different when you have skin in game! Then all that logic stuff doesn’t look so convincing!
I’d just like to take a moment to point out the actual decision that was made here. The judge did _not_ decide it was morally acceptable for minors to have access to Plan B pills. The FDA had already determined that these pills were safe to offer without a prescription. There is no evidence that they have any harmful side effects on minors, or that it is more dangerous for a minor to take them than an adult. Given that, it is prohibited _by statute_ for the FDA to further restrict access without specific scientific evidence of danger – if it’s an OTC med, it has to be treated just like any other OTC med. Period.
The judge ruled, quite properly, that Secy Sebelius’ decision to restrict access to this particular medication was solely political in nature, and therefore was unenforceable.
Doug: “Indeed, even if this ruling stands, it would still be within the authority of Congress to reverse the ruling via legislation that says that minors must have a prescription before obtaining the “morning after” pill. Personally, as I’ve said before, I think all contraceptives, including “morning after” pills should be available over-the-counter, but I do have some doubts about how much that policy ought to apply to minors, especially if we’re talking about girls as young as 13 or 14 years old.”
I would just assume leave that particular issue to the states. According to this link, just over half the states require some form of parental involvement in order for minors to receive contraception services. I assume that the social issues will work themselves out similarly, perhaps with the need for a judicial / medical override if the girl has reason not to inform her parents.
The “rule” I’m sure was only ever added because of pollitical pressure by conservatives. I’m equally sure the FDA are delighted to have their rule over turned..
@stonetools:
Obviously logic isn’t convincing you but to use your own term. At the point a 13 year old needs the morning after pill, the “skin has already been well into the game”.
OK Real World – not RJ logical world:
One one hand, the judge just told the FDA that they have to make a rule in accordance with the scientific evidence, which is pretty much that the Plan B pill can work for any girl who achieves puberty(that’s 11 for many girls these days)
On the other hand, there a lots of parents who don’t want their 13 year old or younger girls buying Plan B over the counter.
And on the gripping foot, Republicans nationwide are going to go apesh*t trying to make political hay out of an “Obamacare” rule saying that 11 or 12 year olds can buy Plan B OTC.
Solution : FDA makes a new rule in accordance with the scientific evidence, but inviting the states to make laws setting parental involvement in the sale of Plan B. Its probably going to suck for Red State teenagers, but them’s the breaks. They should have the good sense to be born where politicians are reality-based.
Compare and contrast.
Seriously ST? Hell slavery sucked for red state slaves too. But hey, we’ll leave it up to the states and if that doesn’t work we’ll allow pharmacists to refuse to provide the pill because it conflicts with their religious fantasies.
@Davebo:
Hey, I’m gaming it out real world, not in the world as I’d like it to be. Above all, I don’t want to hand the Republicans a sexy political issue that they can use to beat the Democrats over the head with in the 2014 elections. You DO remember what happened in 2010 , and how that f**ked things up for Democratic hopes for the rest of the decade? This stuff just ain’t worth a chance to reverse some of that.
If the choice is between the Democrats winning back the House in 2014 and “half-assing” this rule in a way that insulates the Democrats from taking any heat on this, guess what? I’m picking Door No. 1.
@stonetools:
(Ok, I screwed up that post a little (I’m cooking dinner and paying attention to THAT) but you get what I mean.
@stonetools:
You lose that bet.
Um, the skin’s already in the game — or at least in the 13 year old. Once again, at the point a 13 year old girl needs a morning after pill, SHE’S ALREADY HAD SEX, so it’s a little too late to worry about soiling the precious virginal darling with a dirty nasty pill.
Yes, I get what you mean ST.
It’s all politics and if some 14 year old who got knocked up can’t get a morning after pill it’s worth it for the greater goals.
I’m a realist as well but there are some things not worth giving up on for a political advantage. And if it weren’t this do you really believe the GOP couldn’t come up with another wedge issue to toss out to folks who have 13 year old daughters, nieces, etc. who can’t think clearly? (and I’m looking at your here, not the 13 year olds..)
@stonetools:
Do those parents prefer that their 13 year old or younger girls have (a) an abortion or (b) a nine months pregnancy followed by the agonies and dangers of childbirth? Because other than getting lucky and the sperm not implanting, those are the only other options.
I’m betting you’re not a 13 year old girl, right? Bit different when you have skin in the game!
@Davebo: @Rafer Janders:
Here’s the deal. In 2010 Obama appealed to logic, asking the voters to give him another chance and not let the same idiots who drove us into the ditch get back political power. How did that work out for us? So far as I can tell any politician who bases his appeal to voters based on logic risks taking it up the a$$ from the voters.
If you are a political realist you DON’T hand a ready made wedge issue to the Republicans, any more than to hand a loaded gun to a man who’s trying to kill you.
Sorry for those 14 year olds in the red states but we can’t help them unless we win back the House. That’s not how I want it to be: that’s how it IS.
@stonetools: @Rafer Janders: it’s funny. a parent can tell the difference between someone who has kids and someone who doesn’t – just by the way they speak, or write. and you my dear, are no parent. now that doesn’t mean you haven’t procreated, i’m just saying there’s no way you are a parent.
Here’s the the problem i see with your line of thinking – you only see this situation taking place between a boy and a girl of relatively similar ages – 13 seems to be popular, and that is a very romantic thirteen year old thing to think.
but i am a dad. what happens when my 12 year old daughter is molested by the cute old grandpa that lives next door – or a beloved family member that i would never suspect? Virginity is the last thing i could care about. stopping a potentially life threatening pregnancy, testing and treatment of any STD’s – all with ADULT SUPERVISION, not to mention the ability to stop a repeat offense and punish the offender so another child doesn’t suffer. at that age, once is bad enough, repetition can result in physical deformities, MORE mental and emotional damage, and even death. did you know as a parent, in the eyes of the law, i am LEGALLY responsible for my daughters. if they die because of my inaction, i can be punished. but this judge just said i have absolutely no say in what my 12 year old puts into her body? these are high doses of hormones! you know those things we as parents are making a big stink about it being in our food. hormones that can, and do, cause strokes, heart attacks, blood clots, and the list goes on…
i don’t want anyone telling me whats best for my kids. you have kids? fine. then you feed your kids what you want, and i’ll feed my kids what i want. and we will both trust each other’s motives are pure.
@dad:
If the adults in a girl’s life don’t suspect beloved Uncle Lester of being a monster, maybe taking care of her health and figuring out which relatives to trust with her story should be two totally separate questions, and her ability to prevent a pregnancy should not depend on her ability to convince the adults in her life that she’s telling the truth about a beloved family member being a monster.
The poor girl shouldn’t be denied the ability to take care of her health by taking an FDA approved drug because she made a bad call in which adult to trust, should she?
@wr:
I think they need to bring back OTC children’s heroin.
Greetings:
Wasn’t it Hillary Clinton who said, “It takes a government to raise a child.” ???
Children should not be allowed to walk into a drug store and buy just any kind of medicines without parental permission. The drug store around here certainly would not let a child buy medicines without a parent, except for cough drops or Tic Tacs. That is the way it should be everywhere. The schools do not allow medicine at school without parent permission. Certainly 12-16 is way too young for this sort of thing. Most parents around here keep up with their kids.
@dad: but this judge just said i have absolutely no say in what my 12 year old puts into her body?
@Spartacus:
When the judge overturned New York’s ban on sodas, was that judge also saying parents have no say in what their 12 years olds put in their bodies?
@PD Shaw: “I would just assume leave that particular issue to the states”
Here we go again — the absurd fetish with state governments.
For once and for all, could you please explain to me why if the federal government issues the tiniest restrictions, it’s fascism and jackboots. But when a state government decides to regulate the most intimate aspect of our lives, it’s FREEDOM!!!!
If it’s wrong for a government to interfere in our personal lives, why should it make a difference whether the office issuing the rule is two hundred or two thousand miles away?
I really like what you guys tend to be up too.
This kind of clever work and coverage! Keep up the
awesome works guys I’ve included you guys to my own blogroll.
@Tyrell:
MostAll parentsaround hereeverywherekeep up withdon’t have a clue as to what their kids are up to.I am a parent. Children lie to their parents. They keep secrets. It is part of growing up. It is what they do. Think otherwise? Look back to your own childhood. Still think otherwise? You are a fool.
@wr: Is there a single self-styled libertarian out there who believes in freedom for others where it actually inconveniences him? I’ve sure never seen one.
They’re called the ACLU. But for some reason, the glibertarians’ fellow-travelers don’t like them that much. http://www.theonion.com/articles/aclu-defends-nazis-right-to-burn-down-aclu-headqua,1648/
@Rafer Janders:
Do you know many drugs and medications and medical procedures this girl is going to be subject to if she gets pregnant? It’ll make one dose of a morning-after pill look like a sugar cube.
My grandmother gave birth to 12 children without being subjected to any drugs, medications or medical procedures. Same with two of my three kids. The procedures for the third were a choice. You are an idiot.
@Let’s Be Free: Two of your kids gave birth without any prenatal care? That says a lot about the way you live, Mr. Freedom.
@wr:
Not only without any prenatal care, but apparently without any medical procedures, i.e. no childbirth in a hospital with doctors and nurses present.
@Let’s Be Free:
Yes, well, in the year 2013, we do try to offer pregnant teenagers a modicum more medical attention than was available to impoverished backwoods peasants in 1920s Tennessee. Human knowledge marches on, and all that.
@Stormy Dragon:
I screwed up the use of blockquotes. It was Dad – not me – who said parents have no say.
@Tsar Nicholas:
Possible teen pregnancy?
What the heck, another opportunity to create life; and really, aren’t we all pro-life?
Well, that’s nice. Do you know any history beyond family stories? Women died like files in childbirth in the days before modern medicine. Is that what you want to return to?